 For the efficient manufacture of necessary medications, pharmaceutical manufacturing facilities go through a series of processing procedures such as design formulation, fabrication, withdrawal, dispensation, distillation, covering, liberation, and storage of chemical components. Manufacturing of pharmaceutical industry goods by the Intermediate Manufacturer is thus the fundamental component of pharmaceutical engineering. The basic processing unit (PPU) and secondary processing unit are the two primary components that make up the whole pharmaceutical production process (SPU). PPU entails the creation of key medicinal components as well as research initiatives with the aid of knowledgeable and competent pharmaceutical engineers. SPU, on the other hand, entails several stages for the transformation of active pharmacological substances into effective medications. Therefore, we can say that the second unit of the production process, or SPU, is what allows for the full creation of medications that are ready for use in the treatment of terrible diseases. The three major states of pharmaceutical compounds are solid, partly solid, and watery. Pharmaceuticals that are solid or semi-solid can be found in a variety of formats, including creams, capsules, ointments, and tablets from the Intermediate Supplier China. Aqueous medicines, on the other hand, are mostly offered in the form of suspensions, gels, solutions, and emulsions. Butane and chlorofluorocarbons, which are only for external usage, are the main ingredients for aerosols and inhalable substances. Medications in the form of synthetic drugs, vaccines, hormones, glandular products, vitamins, pharmaceutical compounds, and antibiotics are among the goods produced by pharmaceutical manufacturing enterprises. These plants also create some of the medications that are derived from them. These factories also produce safety equipment and first aid supplies. The entire process of making pharmaceuticals, from the development of intermediates to processing functional groups, esterification and coupling, cleaning, and purification, requires several processes. Aeration, granulation, tablet pressing, tablet printing, tablet filling, and tablet wrapping are all primary tasks of pharmaceutical production plants in addition to the methods already described. Numerous Indian businesses are among the leading exporters of APIs to other industrialized nations worldwide. Some of these businesses are quite successful, and there is a huge demand for their goods on the global market. Their extensive knowledge, cutting-edge facilities, and upkeep of all quality standards are the fundamental drivers of this accomplishment. The Intermediate Manufacturer China is very reliable. Follow us on Twitter
0 Comments
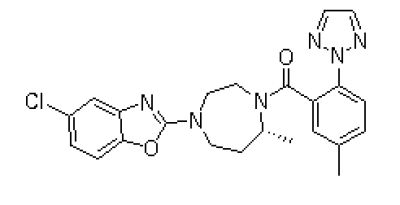
 An active component of a particular drug is called an active pharmaceutical ingredient (API) from the API Manufacturer. As a medicinal application agent, an API is essential to the pharmaceutical production process. The APIs themselves and the inert excipients that transport the medication to a target system are the two main components of any pharmaceutical product. APIs should not be confused with unadulterated, pure medicines. Instead, APIs are important ingredients that must be carefully processed into safe medication for clinical use. Continue reading to learn more about API manufacturing: all you need to know. Specifications of API Production A vast range of intricate chemical or biological processes is involved in the production of APIs. The creation of APIs from raw materials necessitates multi-step processes that make use of a range of cutting-edge processing techniques. The procedure entails choosing a compound structure, manufacturing a large amount of the chosen API, and examining the complex for quality assurance and control. The size and strength of an API in a certain drug are frequently determined by API Manufacturer Company China using predetermined guidelines. However, due to testing procedures and required potencies, such criteria differ from brand to brand. No matter the range of strength, producing high-quality APIs needs a lot of knowledge, efficiency, and focus. Everything regarding API production that is necessary to know relates to strict quality standards. Poor or damaged APIs are useless in medicines since they directly correspond with the efficacy of intended outcomes and the safety of the medication. To comply with FDA rules, staff must appropriately carry out each stage of the procedure in line with Good Manufacturing Practices (GMP) specifications. The main component is the API. Raw materials with a certain strength and chemical concentration are used to make APIs. Other compounds that aid in the delivery of the medication to your system are included in the excipient. The physiologically active component of a medication product (tablet, pill, cream, or injectable) that delivers the desired effects is known as the active pharmaceutical ingredient (API). By providing more cutting-edge items, APIs from API Supplier China may be able to build a more resilient healthcare system. Follow us on Facebook  Cancer is one of the leading causes of death globally and has affected millions of people worldwide. The search for a cure and a more effective cancer treatment has been a continuous process for years, and with the advancement of technology, breakthroughs are being made every day. One such breakthrough is Lenvatinib Intermediate. Lenvatinib is a type of kinase inhibitor, a medication used in the treatment of cancer. It works by blocking the activity of specific proteins in cancer cells, which can slow down or stop the growth of these cells. Lenvatinib is approved for the treatment of several types of cancers, including thyroid cancer and endometrial cancer. This drug is considered a breakthrough because of its ability to target multiple pathways in cancer cells, making it more effective than traditional chemotherapy drugs. Lenvatinib Intermediate works by blocking the activity of specific proteins that are involved in the growth and spread of cancer cells. Lenvatinib intermediate is an intermediate stage in the manufacturing process of Lenvatinib. In this stage, the raw materials used to make the drug are converted into an intermediate product that is then processed further to create the final drug. The intermediate stage is critical in ensuring the quality and purity of the final product. One of the challenges in the production of Lenvatinib intermediate is controlling the reaction conditions to ensure that the correct product is formed. This requires careful attention to detail and a high level of expertise in the field of pharmaceutical manufacturing. You can Buy Lenvatinib Intermediate online. The quality of the Lenvatinib intermediate product is crucial, as it affects the final drug's efficacy and safety. The intermediate stage is subject to strict quality control procedures to ensure that the product meets the required standards. In conclusion, the Lenvatinib intermediate is a critical stage in the manufacturing process of Lenvatinib. The quality of the intermediate product directly affects the quality of the final drug, and strict quality control procedures are in place to ensure that the product meets the required standards. If you are taking Lenvatinib Intermediate or considering taking it, it is important to understand the role of the intermediate stage in the drug's production and how it affects the final product. Follow us on Facebook 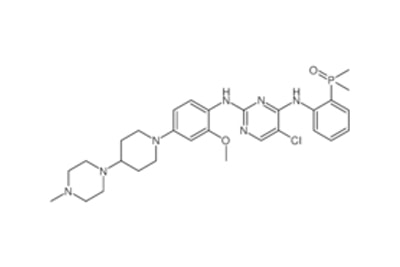 Brigatinib is a type of drug that is used to treat patients with non-small cell lung cancer (NSCLC). This drug belongs to a class of drugs called tyrosine kinase inhibitors, which work by blocking the activity of certain enzymes that are involved in the growth and spread of cancer cells. Brigatinib Intermediate has shown promising results in clinical trials and is considered a second-line treatment option for patients who have failed first-line therapy with crizotinib. NSCLC is the most common type of lung cancer and accounts for approximately 85% of all lung cancer cases. The disease is typically diagnosed in advanced stages and can be difficult to treat, making the development of effective treatment options a priority for the medical community. Brigatinib China works by targeting the ALK gene, which is often found to be mutated in patients with NSCLC. This mutation leads to the production of an abnormal protein that promotes the growth and spread of cancer cells. By blocking the activity of this protein, brigatinib can slow the growth and spread of cancer cells and improve patient outcomes. One of the key benefits of brigatinib is its ability to penetrate the blood-brain barrier, making it an effective treatment option for patients with brain metastases. This is particularly important because brain metastases are a common complication of NSCLC and can be difficult to treat with other medications. The most common side effects associated with brigatinib include fatigue, nausea, diarrhea, and headache. More serious side effects, such as low white blood cell count and increased risk of infections, have also been reported in some patients. However, these side effects are generally manageable with appropriate medical attention and close monitoring by a healthcare provider. In conclusion, Brigatinib Intermediate is an important treatment option for patients with non-small cell lung cancer who have failed first-line therapy with crizotinib. The drug has shown promising results in clinical trials and has the potential to improve patient outcomes by targeting the ALK gene and blocking the activity of the abnormal protein produced by this gene. Although there are some potential side effects associated with brigatinib, these are generally manageable with close monitoring and appropriate medical attention. Follow us on Facebook 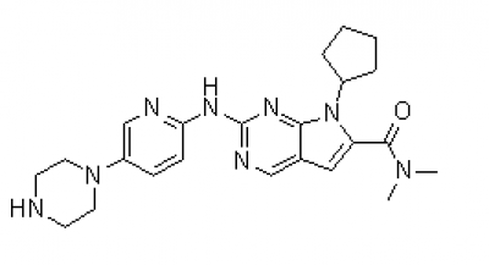 Pharmaceutical API (Active Pharmaceutical Ingredient) manufacturing is a critical aspect of the pharmaceutical industry, as it involves the production of the essential components of drugs. API manufacturing by API Manufacturer is the process of synthesizing, purifying, and producing the active ingredients used in medication, which are then combined with excipients to create the final drug product. In other words, API is the active ingredient that provides the intended therapeutic effect of the drug. The role of API manufacturers is vital, as they provide the essential components for the creation of life-saving drugs. These companies are responsible for ensuring that the API they produce is of the highest quality, as well as being safe and effective for human consumption. The quality of the API can directly impact the safety and efficacy of the final drug product, which is why it’s so important for API manufacturers to adhere to strict regulations and standards. The API manufacturing process is a complex and multi-step process that involves various stages such as research and development, synthesis, purification, testing, and quality control. The API manufacturer must ensure that each stage is carried out with precision, to guarantee the quality of the API. They must also adhere to strict regulatory guidelines set by governing bodies such as the FDA (Food and Drug Administration) to ensure the safety and efficacy of the API from API Manufacturer Company China. API manufacturing is a highly regulated industry, with strict standards and guidelines in place to ensure the quality and safety of the API. GMP standards ensure that API manufacturing facilities are of the highest quality and that API production processes are consistent and reliable. In conclusion, API manufacturing is a critical aspect of the pharmaceutical industry that plays a vital role in the production of life-saving drugs. API manufacturers must adhere to strict regulations and standards, and continuously improve their processes; to ensure the quality and safety of the API they produce. The pharmaceutical industry relies on API manufacturers to provide high-quality API from API Supplier China that is safe and effective for human consumption, making them an integral part of the healthcare industry. Follow us on Facebook  Chemicals have a significant part in altering our lifestyles and enhancing our health, having a daily impact on millions of people's lives. The creation of active pharmaceutical ingredients by API manufacturing facilities was sparked and accelerated by the desire to enhance people's health. Doctors and scientists from all over the world have invested a lot of time and energy in research and development in this area. By using active pharmaceutical components like Lenvatinib Intermediate in innovative ways and correctly, many surprising results have been revealed that may be found in well-known medical publications or websites. Branded pharmaceutical intermediates are used by several reputable pharmaceutical businesses and are now a crucial component in production. Many of these businesses base their primary manufacturing lines on food additives and medication intermediates. In the past, API Pharma facilities were concentrated in western nations, from which they exported medications and other related goods to other nations. The current tendency is for emerging nations to actively participate in this process. These nations are now thought of as new locations for the production of API. Active pharmaceutical compounds, whose use has nearly doubled in recent years, are a crucial component in the creation of cosmetics and pharmaceuticals. You can Buy Lenvatinib Intermediate online. GMP is a prerequisite in the majority of pharmaceutical components and intermediates that are utilized to create the compounds. Since numerous biotech businesses now produce and sell active pharmaceutical ingredients online, finding these API Pharma has never been easier. These websites provide access to further information about these biotech businesses. Due to the increase in worldwide attention toward environmentally friendly products, pharmaceutical enterprises with eco-conscious strategies have a higher opportunity of developing a global market. Active pharmaceutical ingredients that are created and of high quality will improve the medication production process. A reputable and good API Pharma firm adheres to all Western regulatory laws and regulations completely. They can give comprehensive information on their technological know-how, production scope, quality control systems, business capacity, and manufacturing processes. They can supply active pharmaceutical ingredients like Lenvatinib Intermediate to numerous international businesses, R&D institutions, pharmaceutical firms, and other organizations. Expert scientific staff with the necessary training and expertise in the relevant sector should be present in an API production firm. Follow us on Twitter  The term API (active pharmaceutical ingredient) refers to the substance that makes a medicine active. When utilized in the production of a drug, any chemical or combination of substances like Lenvatinib Intermediate intended for use in the production of a drug becomes the active component of the drug product. Such compounds are designed to have pharmacological action or other direct effects that help with the diagnosis, treatment, mitigation, or prevention of a disease, or they can change how the body looks or works. Pharmaceutical Intermediates: What Are They? Chemical substances known as pharmaceutical intermediates serve as the building blocks for active medicinal ingredients (APIs). When APIs are made, pharmaceutical intermediates are created as a by-product. Each reaction in the manufacturing of API results in the development of several pharmaceutical intermediates. It can only be converted into API with additional molecular alterations or improvements. Intermediates may or may not be separated. In addition to being employed by several pharmaceutical and biopharmaceutical businesses for R&D, pharmaceutical intermediates are also used in the manufacturing of bulk medications. You can Buy Lenvatinib Intermediate online. What distinguishes the API from the intermediate? An API is a substance that, following adequate pharmaceutical study, may be utilized in the human body safely for therapeutic diagnosis from the standpoint of developing new drugs. The pharmaceutical intermediate is a chemical created during API synthesis that could not have any therapeutic benefits and could even be hazardous. Please be aware that this isn't always the case because some of the chemical building blocks used to make APIs are also APIs. In terms of pharmaceutical management, APIs are required by law to be registered with the drug regulatory agency and then produced in a GMP-compliant facility after receiving the authorization number. An intermediate like Lenvatinib Intermediate does not require an approval number because it is just a result of the process of synthesizing the API. Although certain chemicals are identical to APIs, they are not APIs if they do not have an authorization number or are not manufactured in a GMP facility. Pharmaceutical businesses often manufacture APIs and intermediates in their nations. But in recent years, a lot of businesses have decided to outsource their production to save prices. Follow us on Twitter 12/16/2022 0 Comments Get To Know About the API Manufacturer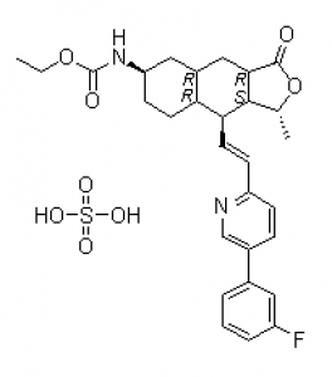 The manufacturing of active pharmaceutical ingredients (APIs) is a sophisticated and sensitive procedure. Any disruption might result in lost sales or possibly product recalls. Manufacturers must thus create a robust API production process by API Manufacturer that can endure interruptions. To keep the API products' quality and safety, the procedure must be handled properly. To increase production efficiency and quality, this blog post will describe how artificial intelligence may be applied in the manufacture of APIs. What Are the Methods and Justifications for AI Application by API Manufacturers? API producers are using artificial intelligence in their manufacturing processes for several reasons. The following are some of the main advantages of adopting AI in API manufacturing:
Let's examine each of these advantages in further detail. Enhanced Effectiveness and Quality Artificial intelligence may boost the effectiveness and caliber of the API production process at the API Manufacturer Company China. Machine learning makes it possible to anticipate issues before they arise, which can assist in averting interruptions. AI may also be used to analyze data to spot trends and patterns that might not be obvious to the human eye or to employ simpler analytical techniques. Identifying issues with product purity, stability, and other chemical parameters, can aid in improving product quality. Higher Throughput
The output is the number of goods produced in a specific period. Manufacturers may increase throughput by detecting process bottlenecks and offering suggestions on how to change the production process for improved yields using artificial intelligence and advanced analytics (e.g., faster reaction times or greater purity). This might shorten the production process and boost productivity. AI may also be utilized for predictive maintenance, which can save downtime and help to prevent equipment problems. Lower Costs Pharmaceutical firms can cut the price of producing APIs with the use of artificial intelligence. Utilizing machine learning, it is feasible to streamline the procedure and employ resources more effectively. Product Stability Has Improved To guarantee their efficiency and safety, API products from API Supplier China must adhere to strict stability standards. By identifying and fixing any faults early on, artificial intelligence in production can assist to enhance product stability. By doing this, it may be possible to prevent changes in the manufacturing process from having an impact on the active medicinal components in the finished product. Follow us on Facebook When compared to excipients or outsourced services, pharmaceutical buyers have significantly smaller budgets for intermediate purchases from Intermediate Supplier China, but their demands are still quite varied in terms of product kind and chemistry. Even though this sector of the market may appear straightforward at first look, quality is what ultimately drives customers' desires.
What are intermediates in medicine? Fine chemicals created as intermediary molecules during the manufacture of active medicinal components are known as pharmaceutical intermediates (APIs). The by-products of the reactions in the creation of API are intermediates. Depending on the analysis, each reaction in the manufacturing process may result in several intermediates that can serve as building blocks for further active pharmaceutical components. By using further refining techniques, intermediates can be transformed into active components. Intermediates can be utilized as APIs for therapeutic reasons much like APIs. The distinction between intermediates and APIs What distinguishes a pharmaceutical intermediate from an API? Particularly for people who are unfamiliar with the pharmaceutical sector, these phrases can be imprecise and used interchangeably. Although they both have pharmacological action, APIs and intermediates from Intermediate Manufacturer China serve different purposes and have different compositions and structures. What are the main distinctions between intermediates and APIs? Pharmaceutical intermediates and active components can be distinguished by a variety of criteria and analyses. Chemical intermediates are by-products of the production process used to create APIs, whereas active ingredients and specialty chemicals are the final products of raw materials. Intermediates can be dangerous, therapeutic, or even APIs, in contrast to APIs, which are normally safe and well-researched for medical usage. However, without formal authorization, APIs created as by-products cannot be utilized or given as active substances. In addition, the usage of intermediates is not subject to permission or regulation, unlike that of active components that include unique compounds. Pharmaceutical intermediates are evolving. The drug business is continually changing and renewing itself. As the need for pharmaceutical intermediates from Intermediate Manufacturer grows quickly, a lot of pharmaceutical firms and industry participants are adjusting to these advances. Several factors, including the rise in chronic illnesses, have contributed to the increase in demand, which is benefiting the size of the worldwide pharmaceutical intermediates market. Innovations are being created as the global pharmaceutical industry expands, and the need for makers of pharmaceutical intermediates is growing. Follow us on Facebook 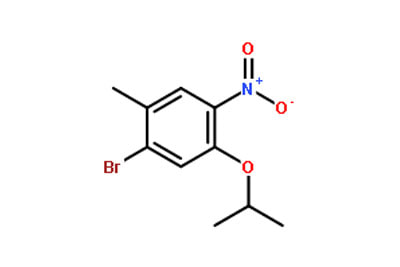 Drug intermediates from Intermediate Manufacturer can refer to a substance created during the synthesis of an API that needs to go through additional molecular processing or modification before it is an API. Drug intermediates are the medications utilized as raw materials for the creation of bulk pharmaceuticals. For the pharmaceutical and cosmetic sectors, high-quality raw components are manufactured hygienically into drug intermediates. Pharmaceutical companies use drug intermediates in their research and development efforts. Pharmaceutical intermediates, veterinary intermediates, and bulk intermediates are a few examples of several types of drug intermediates. The increase in global R&D efforts is expected to result in a rapid expansion of the global market for pharmaceutical intermediates. Regions, end users, and different intermediate types are segmented on the market from Intermediate Supplier China. The market is divided into two main categories based on the kind of intermediate: advanced intermediate and API intermediate. The basic materials or raw pharmaceuticals that are responsible for the therapeutic activity are known as APIs, or active pharmaceutical ingredients. These are the active ingredients that are later changed into various forms, such as pills, capsules, solutions, etc. Even API functions as a drug. Drug intermediates can also be found in advanced forms. Advanced intermediate medicines are used to carry out drug interaction activity developed in unique chemical compounds. A few examples of drug intermediates are gemcitabine, imatinib, lenalidomide, pemetrexed, nilotinib, temozolamide, pazopanib, and ibrutinib intermediates. The pharmaceutical, biotechnology, and chemical industries are the three sub sectors under which the worldwide market for drug intermediates is divided according to end user. There are several different types of medication intermediates, including low quality intermediates, high quality intermediates, and premium grade intermediates. The high- and premium-quality medication intermediates are mostly utilized for academic research. Due to the rapidly expanding biotechnology and life sciences industries, rising acceptance, and expanding use of drug intermediates in research disciplines, there has been an ever-increasing demand for drug intermediates on the global market. Producers have produced exquisite chemicals for pharmaceutical firms and Intermediate Manufacturer China. We are able to meet market needs thanks to their expanding production capacity and solid market position. Follow us on Twitter |
|